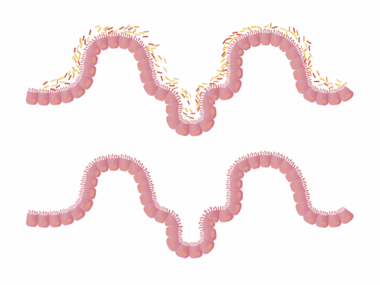
The Impact of Antibiotics on Your Gut and Immune Defense
Antibiotics are often prescribed to combat bacterial infections, but their impacts extend beyond just eradicating harmful bacteria. They can also significantly disrupt the delicate balance of beneficial microorganisms residing in the gut. These microbes play an essential role in digestion, nutrient absorption, and maintaining overall gut health. When antibiotics are taken, they indiscriminately kill both harmful and beneficial bacteria, leading to conditions such as dysbiosis. Dysbiosis can increase susceptibility to gastrointestinal issues like diarrhea, bloating, and even irritable bowel syndrome. Furthermore, the loss of healthy gut bacteria can weaken the immune system’s performance. The gut houses approximately 70% of the immune system, so its health is intimately linked to overall immunity. Low levels of beneficial bacteria can impair the body’s ability to fight off infections, leading to increased chances of illness. Therefore, while antibiotics serve an important purpose, understanding their impact on gut health is crucial. Probiotics and dietary measures can help restore balance after antibiotic treatment, supporting both gut function and immune defense. Consulting healthcare professionals for proper antibiotic use and complementary therapies is advisable.
Understanding Gut Microbiota
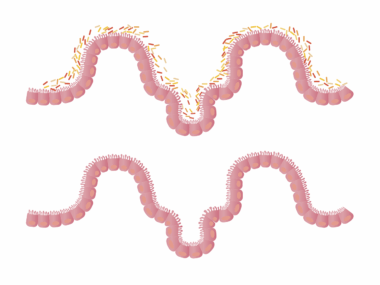
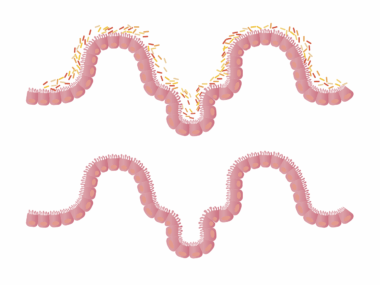
The gut microbiota, a complex community of trillions of microorganisms, is crucial for our health and well-being. This community includes bacteria, viruses, fungi, and other microbes that collaborate to perform various functions. These beneficial microbes aid digestion, synthesize essential vitamins, and protect us against pathogenic bacteria. In fact, diverse gut microbiota is linked to better health outcomes and improved immune responses. However, antibiotic use can dramatically alter the composition and diversity of gut microbiota, typically reducing beneficial bacteria while allowing opportunistic pathogens to flourish. This imbalance can create a breeding ground for infections and other health complications. Research has shown that an altered microbiome may also contribute to conditions such as obesity, allergies, and autoimmune disorders. To help mitigate the negative effects of antibiotics on gut microbiota, individuals are encouraged to consume a diet rich in prebiotics and probiotics. Foods like yogurt, sauerkraut, and fiber-rich vegetables can foster the replenishment of beneficial gut microbes. Keeping the gut microbiota healthy is not only vital for digestion but is also essential for robust immune function and overall health.
While antibiotics are often necessary for treating infections, their impact on gut health and immunity cannot be overlooked. Following a course of antibiotics, many individuals may experience gut disturbances that signpost reduced gut flora diversity. This absence of diverse bacteria can hinder optimal gut functionality, which serves as a frontline defense against pathogens. As the gut is integral to immune response, a compromised gut may lead to increased chances of illness and infections. Studies have shown that individuals who have taken antibiotics repeatedly may be more susceptible to conditions like C. difficile infections, which can cause severe diarrhea and colitis. Additionally, chronic antibiotic use can lead to a long-term alteration of gut microbiota structure, having sustained ramifications on health. This disruption in gut defense can be particularly concerning in young children and the elderly, who may already have vulnerable immune systems. It is essential to balance the necessity of antibiotic use with proactive strategies to maintain gut health, such as dietary interventions. Endorsing appropriate medical advice post-antibiotic treatment is vital for restoring gut health and enhancing immunity over time.
The use of probiotics as a supportive measure during or after antibiotic treatment has gained significant attention in recent years. Probiotics are live microorganisms, often referred to as “good bacteria,” that can enhance gut health and immune function. The rationale for using probiotics is rooted in their ability to help replenish beneficial gut bacteria that may have been lost during antibiotic therapy. Certain strains of probiotics have been found to reduce the incidence of antibiotic-associated diarrhea and may promote a faster recovery of gut microbiota after antibiotic use. In fact, some studies have indicated that probiotics can support the immune system by modulating immune responses, potentially decreasing inflammation associated with gut disturbances. It is essential, however, to choose appropriate probiotic strains that have been clinically tested for efficacy, as not all probiotics are the same. Additionally, it’s advisable to discuss their use with a healthcare provider to ensure compatibility with individual health needs. By incorporating probiotics into recovery strategies, individuals may enhance both gut health and immune resilience post-antibiotic therapy.
Nourishing Your Gut
The role of diet in supporting gut health during and after antibiotic treatment is paramount. Foods rich in prebiotics, which feed beneficial gut bacteria, can help maintain a healthy gut environment. Foods such as garlic, onions, and asparagus provide essential nutrients that encourage the growth and maintenance of good microbes. Fermented foods like kimchi, kefir, and miso are also beneficial as they are natural sources of probiotics. A balanced diet incorporating fruits, vegetables, whole grains, and lean proteins can support not only gut health but also overall immune defense. Additionally, hydration plays a critical role in gastrointestinal health. Consuming adequate water helps maintain proper digestive processes and prevents issues like constipation, which can occur after antibiotic use. It’s advisable to limit processed foods, added sugars, and artificial sweeteners during recovery since these can disrupt healthy gut microbiota. Making conscious dietary choices can fortify the body’s defenses against pathogens and promote a balanced microbiome. Individuals should consider consulting with a nutritionist to develop personalized meal plans tailored to specific health needs immediately after antibiotic treatment.
Stress management also plays an influential role in maintaining gut health and immune function. Chronic stress can negatively impact the gut microbiota, exacerbating the effects of antibiotics on gut health. Stress can lead to increased levels of cortisol, a hormone that can alter gastrointestinal motility and contribute to various digestive disorders. Techniques focused on reducing stress, such as mindfulness, meditation, yoga, and regular physical activity, can have a positive impact on gut health by promoting microbial diversity. Engaging in relaxation techniques provides dual benefits by helping with mental well-being and aiding in the replenishment of beneficial gut bacteria. A positive mindset and a manageable stress level can also enhance immune responses, allowing the body to combat infections more effectively. Furthermore, social support and connections can alleviate stress and contribute to healthier lifestyles. Individuals should aim to combine physical, emotional, and social wellness strategies for optimal gut health, especially after a course of antibiotics. Implementing these practices can create a synergistic approach to improving both gut and immune health sustainably.
Given the importance of gut health in overall immunity, it is crucial to raise awareness about the potential consequences of antibiotic overuse. Education around responsible antibiotic prescribing and consumption can help mitigate the long-term impacts on gut microbiota and immunity. Patients should engage in conversations with healthcare providers about the necessity of antibiotics and alternatives when appropriate. Additionally, understanding the importance of completing prescribed antibiotic courses, while also discussing follow-up care, is beneficial. Future investigations are essential to further understand the intricate connection between antibiotics, gut health, and immune responses. By fostering policies that encourage prudent antibiotic use, we can protect not only individual health but also public health. Raising awareness through community programs and healthcare institutions can enhance knowledge on maintaining gut health before, during, and after antibiotic treatment. Supporting research in this field will potentially revolutionize approaches to antibiotic therapy, encouraging practices that prioritize gut health alongside infection control. Monitoring antibiotic consumption patterns can also serve as a preventative measure against antibiotic resistance, which poses a significant threat to global health.
In conclusion, the relationship between antibiotics, gut health, and immune defense is multifaceted and critical to understand. Antibiotics can disrupt gut microbiota and impact immunity, leading to various health complications. Probiotics, dietary considerations, and stress management are effective strategies for mitigating these effects. As we navigate antibiotic therapies, emphasizing gut health becomes paramount for enhancing immunity and overall well-being. Personal health decisions surrounding antibiotics should be informed by comprehensive knowledge about their effects on gut microbiota health. The continual study of the gut-immune connection will guide future healthcare practices, particularly as we face increasing antibiotic resistance. Supportive measures like nutrition and stress management should be integrated into recovery plans to promote resilience and optimal health. Public health efforts focused on proper antibiotic use can help create a healthier society with stronger immune defenses. The path to understanding this impact will lead to better health outcomes, protection against infections, and improved quality of life. Ultimately, awareness and preparation can empower individuals to make informed health decisions that align with gut health goals and enhance overall immunity.