Gut Microbiota and Inflammatory Bowel Disease: What We Know
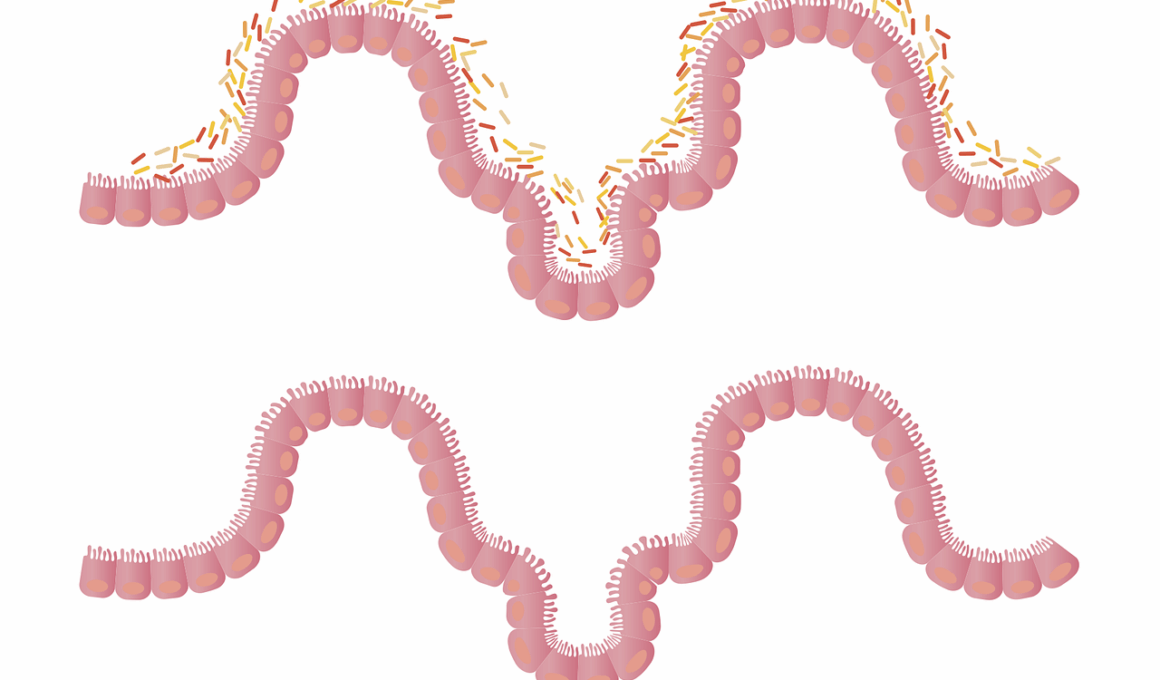
The gut microbiome is a complex community of microorganisms residing in our digestive tract, playing a significant role in various health aspects. Research suggests that an imbalance in these microorganisms may contribute to inflammatory bowel disease (IBD), characterized by chronic inflammation of the gastrointestinal tract. Two primary forms of IBD are Crohn’s disease and ulcerative colitis, both of which disrupt normal gut function. Healthy gut microbiota promotes digestion, synthesizes essential nutrients, and aids in maintaining the integrity of the gut barrier. Conversely, dysbiosis, or microbial imbalances, is associated with increased intestinal permeability and inflammation. The link between gut microbiome and IBD has led to exploring fecal microbiota transplantation as a therapeutic approach, restoring a healthy microbial balance. Understanding the gut-brain axis also provides insight into how mental health affects gastrointestinal health. Future research is vital for uncovering how specific strains of bacteria impact immune responses, potentially informing the development of targeted therapies. The interplay between genetics, lifestyle, and the gut microbiome remains a critical focus in IBD research.
Role of Microbiota in Inflammatory Bowel Disease
In studying IBD, scientists have identified several specific changes in gut microbiota composition. There is a notable decrease in beneficial bacteria such as Faecalibacterium prausnitzii and Ruminococcus, key players in maintaining gut health. Increased levels of pathogenic bacteria, including Escherichia coli and Fusobacterium, have been observed in IBD patients. This alteration is thought to trigger an inappropriate immune response, resulting in chronic inflammation. Furthermore, specific dietary patterns can influence these microbial communities, which highlights the importance of nutrition in managing IBD. By modifying dietary habits to incorporate more fiber and fermented foods, patients may positively impact their gut microbiome. Personalized nutrition plans tailored specifically for IBD patients could enhance therapeutic outcomes and overall well-being. Additionally, probiotics and prebiotics are being studied for their potential benefits in IBD management. While emerging evidence supports their use, more randomized controlled trials are required before making definitive recommendations. Ultimately, understanding the connection between gut microbiota modifications and the development or exacerbation of IBD is an ongoing area of investigation.
The gut-brain axis is an emerging area of research that connects gut microbiota and mental health. Recent studies suggest that the microorganisms in the gut communicate with the brain through various pathways, including the vagus nerve and immune system. This interaction appears to influence mood and behavior, potentially affecting the severity of conditions like IBD. Stress and mental health disorders have been associated with dysbiosis and can exacerbate gastrointestinal symptoms. Understanding this connection may lead to innovative approaches that incorporate mental health support into IBD management. For instance, stress-reduction techniques such as mindfulness, cognitive behavioral therapy, and yoga can offer mental benefits that positively affect gut health. Additionally, researchers are exploring the efficacy of psychoactive antibiotics and medications aimed at improving gut microbiota composition. Such approaches could alleviate symptoms of both IBD and anxiety or depression, improving life quality for patients. Continued exploration into the gut-brain axis is crucial for developing comprehensive treatment strategies. An integrative approach addressing both psychological factors and gut health may yield significant improvements for individuals suffering from inflammatory bowel diseases.
Therapeutic Interventions for IBD
Numerous therapeutic approaches are available for managing inflammatory bowel disease, each tailored to the severity of the condition and individual patient needs. Conventional treatments often include anti-inflammatory medications, immunosuppressants, and corticosteroids. However, these options can come with side effects, leading to the exploration of alternative therapies. Nutritional therapy is gaining traction, with specific diets aimed at reducing inflammation and promoting healing. The Specific Carbohydrate Diet and Low Fermentable Oligosaccharides, Disaccharides, Monosaccharides, and Polyols diet have shown benefits for some patients. Integrating probiotics and prebiotics into daily routines is also being researched as adjunct therapy. These supplements may help restore healthy microbiota balance and reduce flare-ups. Advances in biologic therapies targeting specific immune responses have also transformed the management of IBD, providing options for patients with moderate to severe disease who do not respond to traditional treatments. Further studies are needed to determine the optimal combination of therapies, including the timing and order of interventions, for maximizing patient outcomes and minimizing side effects. Such personalized approaches hold promise for improving IBD management.
Emerging research suggests that the gut microbiome could also be influenced by environmental factors, including antibiotic usage and diet. The overuse of antibiotics can lead to significant disruptions in gut microbial communities, which may not recover to their original state, potentially predisposing individuals to autoimmune diseases, including IBD. Additionally, Western dietary patterns high in fats, sugars, and processed foods can adversely affect the gut microbiota. Such diets can promote inflammation and disturb microbial diversity. On the other hand, incorporating a Mediterranean-style diet rich in fruits, vegetables, whole grains, and healthy fats appears to foster a more favorable gut microbiome. The potential reversibility of gut dysbiosis raises hope for future interventions in IBD. Ongoing investigations are focusing on how introducing specific dietary components or probiotics can positively influence microbial composition and improve gut function. Understanding the impact of our environment and lifestyle on gut microbiota is essential in formulating preventative strategies for IBD and possibly other autoimmune disorders. Adopting lifestyle changes may empower individuals to influence their gut health positively, demonstrating the exciting potential of dietary interventions.
The Future of Gut Microbiome Research
The future of gut microbiome research holds promise for innovative treatments and preventive strategies for inflammatory bowel disease. With advancing sequencing technologies and bioinformatics, researchers can analyze the microbial landscape in unprecedented detail. Big data analytics and machine learning may soon play a critical role in predicting individual responses to specific therapies based on gut microbiota composition. This personalized medicine approach can revolutionize treatment protocols, leading to improved patient outcomes. Furthermore, ongoing studies are investigating the efficacy of bacteriophages as therapeutic agents in combating dysbiosis. These viruses target specific pathogens, thereby sparing beneficial bacteria and restoring healthy microbial balance. Clinical trials focusing on synbiotics, a combination of probiotics and prebiotics, are also underway, aiming to optimize gut health in IBD patients. Ongoing collaborations across disciplines, including microbiology, immunology, and nutrition, will enhance our understanding of the intricate relationships between gut microbiota and autoimmune diseases. Ultimately, a holistic understanding of the gut microbiome’s role in health and disease will pave the way for novel therapeutic avenues. As research continues to evolve, we hope researchers will illuminate the pathways involved in gut dysbiosis and inflammation.
In conclusion, the gut microbiome plays a crucial role in the pathogenesis and management of inflammatory bowel diseases. By understanding these intricate relationships between gut bacteria and host immunity, innovative therapeutic approaches are emerging. Diet and lifestyle modifications, probiotics, and prebiotics hold significant potential for restoring a healthy gut microbiota balance. Continued research is essential in deciphering this complex interplay so that healthcare providers can effectively address the multiple aspects of IBD management. As science uncovers more about the microbiome, patients could benefit from personalized interventions tailored to their unique microbiota profiles. It is an exciting time in this field, offering newfound hope and strategies. Emphasizing a multifaceted approach that includes psychological support and dietary modifications may also enhance patient outcomes. The complex relationship between gut health, immunity, and the brain must not be overlooked, as it informs comprehensive care strategies. In the future, monitoring gut microbiota could become vital in diagnosing, treating, and preventing autoimmune diseases like IBD. The potential for microbiome-focused therapies could profoundly change the landscape of inflammatory bowel disease management.
Understanding the various factors influencing gut microbiota composition and the resulting health implications allows for better management of autoimmune conditions. The gut microbiome’s dynamic nature underscores the importance of ongoing research in this area. Researchers are investigating how genetic predispositions, environmental exposures, dietary habits, and lifestyle choices combine to shape the gut microbiome. Uncovering these connections can enable healthcare practitioners to design personalized treatment approaches that resonate with individual patient needs. This personalized medicine approach may lead to more effective management strategies in inflammatory bowel disease treatment. As we look ahead, the integration of tools for microbiome analysis into clinical practice will be essential for assessing gut health over time and its impact on autoimmune diseases. In summary, the connection between gut microbiota and inflammatory bowel disease presents exciting prospects for advancing therapeutic strategies. As insights deepen, researchers and clinicians can work collaboratively to unravel the complexities of the gut microbiome and its pivotal role in autoimmune health. Patients equipped with this knowledge can be empowered to make informed lifestyle choices to support a healthier gut and potentially mitigate the impacts of their conditions.