Gut Microbiome Changes Triggered by Antibiotic Usage
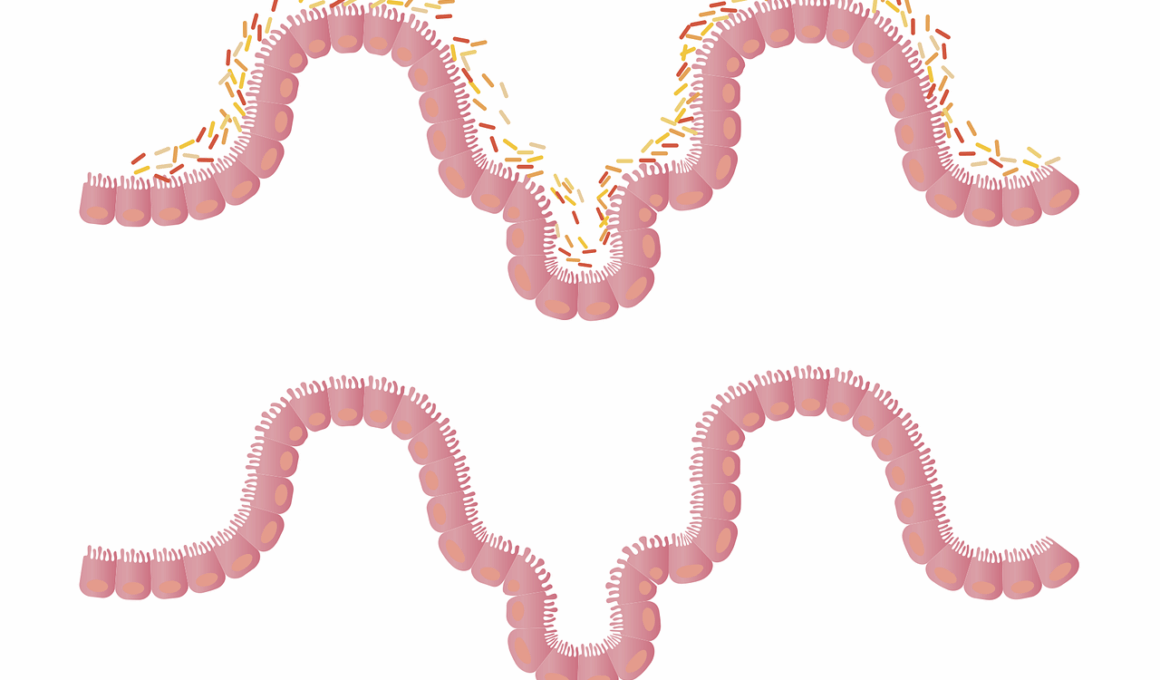
The gut microbiome consists of trillions of microorganisms that inhabit the digestive tract, playing essential roles in human health. Antibiotics are potent medications that can disrupt this delicate microbial balance. While antibiotics are designed to treat bacterial infections, they can also kill beneficial bacteria alongside the harmful ones. The resulting disruption leads to dysbiosis, a significant imbalance in the gut microbiota. Several factors influence the extent of changes in the gut microbiome due to antibiotic usage. The type and duration of the antibiotic course are crucial, with broader-spectrum antibiotics generally causing more significant alterations. Furthermore, individual factors such as age, diet, and genetic predisposition also play important roles in how the gut microbiome responds. This disruption can manifest as various gastrointestinal issues, including diarrhea, abdominal pain, and bloating, adversely affecting overall health. Understanding these changes is vital for both healthcare providers and patients to mitigate the adverse consequences of antibiotics. Consequently, alternative treatments and probiotic therapies may be recommended to restore the gut microbiome post-treatment. By maintaining gut health, we can improve our overall well-being and reduce the negative effects of antibiotic usage.
Consequences of Antibiotic-Induced Dysbiosis
Antibiotic-induced dysbiosis contributes to a range of health issues beyond gut disturbances. A compromised gut microbiome can weaken the immune system, making individuals more susceptible to infections and diseases. Beneficial gut bacteria are essential for synthesizing vitamins, metabolizing nutrients, and reinforcing the gut barrier against pathogens. Disruption of these functions can lead to nutrient deficiencies, increased inflammatory responses, and systemic health problems. Moreover, alterations in the gut microbiota can affect mental health, linking gut health to psychological well-being. This connection is often referred to as the gut-brain axis. Research suggests that an imbalanced microbiome may contribute to mood disorders, anxiety, and cognitive dysfunctions. Restoring gut health after antibiotic treatment is crucial in reversing these adverse effects. Probiotics, prebiotics, and dietary changes can support the recovery of beneficial microbes. It’s also vital to approach antibiotic usage judiciously, reserving them for confirmed bacterial infections and avoiding unnecessary prescriptions. Patients must communicate openly with healthcare providers about their antibiotic history to tailor appropriate microbiome recovery strategies and preserve gut health.
The Role of Probiotics in Recovery
Probiotics are live microorganisms that provide health benefits when consumed in adequate amounts. They have been widely studied for their potential to restore gut microbiome balance following antibiotic therapy. Several studies have demonstrated that probiotics can help mitigate antibiotic-associated diarrhea and other gastrointestinal disturbances. By replenishing beneficial bacteria, probiotics may help the gut regain its natural diversity and functionality. Different probiotic strains may have varying effects, making it essential to choose specific strains targeting particular health issues. For example, Lactobacillus and Bifidobacterium species are commonly recommended for their antimicrobial properties and health-promoting effects. In clinical settings, healthcare providers may counsel patients on incorporating probiotic-rich foods like yogurt, kefir, and fermented vegetables into their diet. Additionally, probiotic supplements are available for those seeking more concentrated doses. While probiotics can significantly aid recovery, individual responses may vary. It is vital to continue monitoring gut health through dietary adjustments and lifestyle modifications to enhance the benefits of probiotics and achieve lasting balance in the gut microbiome following antibiotic use.
The timing of probiotic administration is crucial for maximizing their benefits. Taking probiotics simultaneously with antibiotics may reduce their efficacy due to the antibiotics also targeting the introduced bacteria. Therefore, it is generally recommended to consume probiotics at least two hours after antibiotic doses. This timing allows beneficial bacteria to withstand the impact of antibiotics while also promoting growth and colonization in the gut. Additionally, maintaining a balanced diet rich in prebiotics can further support gut health. Prebiotics are non-digestible food components, mostly found in high-fiber foods, which nourish beneficial gut bacteria. Examples include bananas, onions, garlic, and whole grains. Combining probiotics and prebiotics can enhance their effects, a strategy known as synbiotics. It is crucial for individuals recovering from antibiotic therapy to be proactive about their gut health. Monitoring dietary intake and making conscious choices can significantly contribute to restoring the microbiome’s equilibrium. Engaging with healthcare professionals about optimal probiotic and dietary interventions can also empower patients in managing their gut health, ensuring their recovery process is as effective as possible.
Emerging research continues to explore the long-term impacts of antibiotic use on the gut microbiome. Some studies suggest that significant disruptions may persist for months or even years after the completion of a course of antibiotics. The implications are profound, particularly considering the rising trend of antibiotic misuse and over-prescription. The development of antibiotic-resistant bacteria is a growing concern directly related to this issue. Such resistance can not only exacerbate health problems but also complicate treatment strategies. As a result, there is an urgent need for continued public education regarding the responsible use of antibiotics. Health organizations recommend improving awareness of appropriate antibiotic use, focusing on when they are necessary and how to lessen their impacts. Implementing guidelines on antibiotic stewardship can help minimize unnecessary prescriptions. This approach is vital for preserving gut microbiome health and preventing the negative consequences of antibiotic-induced dysbiosis. Patients should advocate for their health by discussing concerns with healthcare providers and exploring alternative treatment options whenever possible to protect their microbiome and overall health.
Dietary Strategies to Support Gut Health
Substantial evidence supports the role of diet in shaping the gut microbiome. A well-balanced diet rich in whole, minimally processed foods is essential for sustaining a healthy microbiome. Foods high in dietary fiber, such as fruits, vegetables, legumes, and whole grains, serve as fuel for beneficial gut bacteria. Incorporating diverse food sources encourages microbial diversity, which is integral for overall gut health. The Mediterranean diet, known for its abundance of plant-based foods, healthy fats, and lean protein sources, has been associated with improved gut microbiota composition. Avoiding excessive sugar, unhealthy fats, and processed foods is equally important, as these can promote the growth of harmful bacteria and contribute to dysbiosis. Hydration is also crucial, as water supports digestion and gut barrier function. In contrast, high consumption of antibiotics may necessitate dietary vigilance. Nutritional interventions can mitigate disruption. Individuals can benefit from pooling knowledge of gut-health-promoting foods to implement dietary strategies that best fit their lifestyle. Collaborating with nutritionists can further optimize their approach to supporting gut health following antibiotic usage.
Incorporating fermented foods into the diet is another highly recommended strategy for enhancing gut health. Fermented foods are rich in probiotics, creating a diverse range of beneficial bacteria. Examples of these foods are kimchi, sauerkraut, kombucha, and miso, all known for their gut-friendly properties. Regularly consuming these items can help restore balance to the microbiome and provide a range of health benefits. Additionally, adopting cooking methods that enhance nutrient availability can further support gut health. Steaming, roasting, or fermenting can make beneficial compounds in food more accessible, contributing to better overall digestion and absorption. Balancing gut health extends beyond individual choices, as it can be influenced by lifestyle factors, such as stress and sleep patterns. Stress management techniques, including mindfulness and physical activity, can positively impact the gut-brain axis. Adequate sleep is essential for overall health, including metabolic health which ties into gut balance. Recognizing the interconnectedness of physical and mental well-being facilitates a comprehensive approach to maintaining gut health post-antibiotic usage. By employing multidisciplinary strategies, individuals can foster a healthier gut microbiome and improve quality of life.
The Future of Gut Health Management
As research on the gut microbiome progresses, innovative approaches for gut health management are emerging. Future studies will likely delve deeper into individual microbiomes, leading to personalized interventions tailored to specific health needs. Advances in microbiome analysis technology may allow for more precise assessments and recommendations regarding diet and probiotic use. Additionally, the potential for fecal microbiota transplantation (FMT) is being explored as a treatment option for severe dysbiosis, especially in cases where traditional methods have failed. While still a developing field, FMT shows promise for restoring a balanced microbiome and improving health outcomes. Establishing robust guidelines on antimicrobial substance usage is paramount for preserving gut microbiome health amidst the challenges posed by antibiotic resistance. Public health initiatives will need to emphasize preventive strategies across communities. Nurturing healthy gut microbiomes becomes imperative for individual wellness and public health. Combining scientific research with practical guidelines will allow individuals to make informed decisions about their gut health. A proactive approach, rooted in collaboration between patients, providers, and researchers, can pave the way for better management of gut microbiome health in the future.