Gut Microbiome in Aging Populations: Cancer Prevention Implications
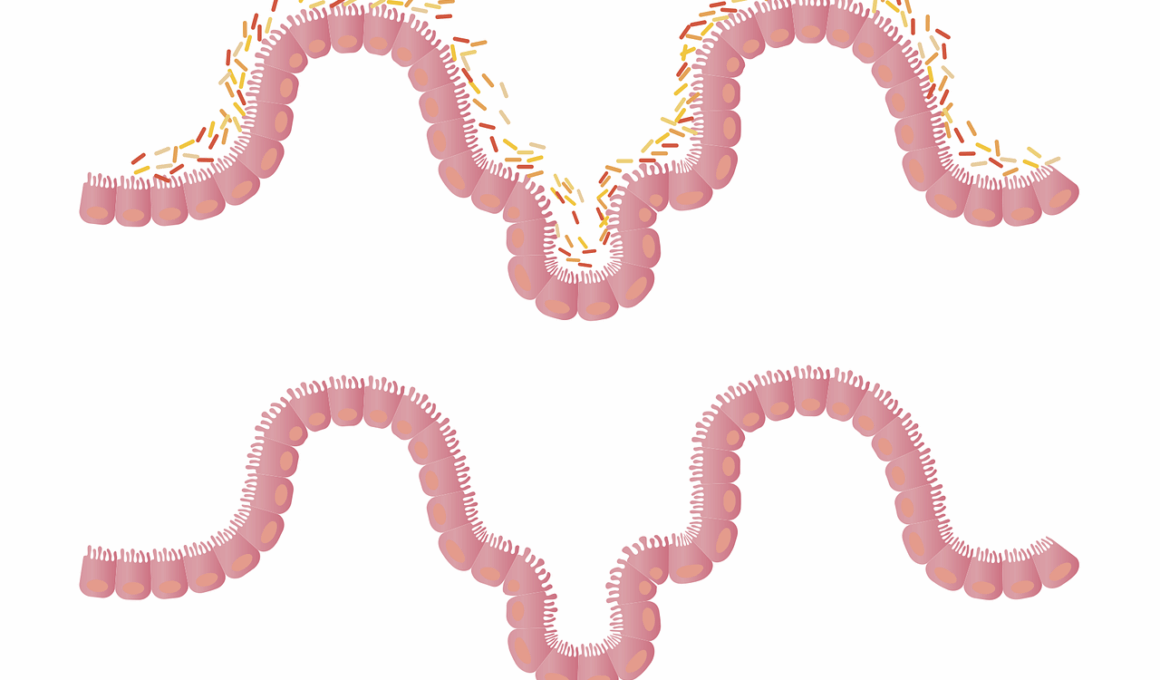
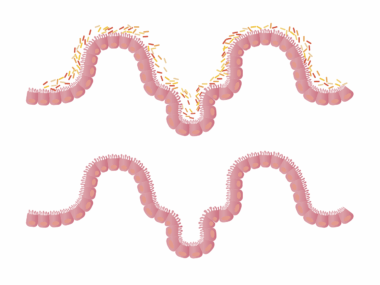
The human gut microbiome is a diverse ecosystem comprising trillions of microorganisms, influencing various aspects of health. In aging populations, this microbial community undergoes significant changes, which can have substantial implications for cancer prevention. As we age, the diversity and composition of gut bacteria tend to decline, leading to dysbiosis, a condition linked with increased cancer risk. Factors such as diet, medications, and lifestyle choices can exacerbate these changes. Research indicates that a balanced microbiome supports a robust immune system and may help in the detection and elimination of cancerous cells. In particular, the presence of certain beneficial bacteria has been associated with reduced inflammation and better metabolic health, both of which are vital in lowering cancer risk. Therefore, maintaining gut health through dietary interventions and probiotics is crucial in promoting longevity and potentially preventing cancer. A multifaceted approach that emphasizes personalized nutrition and lifestyle modifications can optimize gut microbiome health in older adults. Continued exploration of this area is essential for developing effective strategies in cancer prevention for aging populations.
Changes in the gut microbiome can significantly affect cancer associated with age. This phenomenon is particularly critical in older adults, whose microbiomes often exhibit reduced diversity and beneficial bacterial populations. Resilience against pathogens is weakened, allowing harmful bacteria to proliferate, creating an inflammatory environment conducive to cancer development. Furthermore, metabolites produced by gut microbes, such as short-chain fatty acids (SCFAs), play pivotal roles in maintaining colonic health and regulating immune responses. Studies have demonstrated that an increase in SCFAs can inhibit tumorigenesis in the colon. Interventions aimed at restoring diversity in the microbiome through diet or probiotics have shown promise in cancer prevention efforts. Foods rich in fiber, such as fruits, vegetables, and whole grains, can nurture a favorable microbiome composition. These dietary choices not only enhance microbial diversity but also promote the production of protective metabolites against cancer. Additionally, understanding the interplay between gut microbiota and other factors, such as genetics and environmental influences, is integral. This complex relationship holds the key to developing targeted therapies and prevention strategies aimed at mitigating cancer risks in aging populations.
The Role of Diet in Modulating Gut Microbiome
The role of diet in modulating the gut microbiome cannot be overstated, particularly in older adults at risk of cancer. Dietary patterns rich in fiber, fruits, and vegetables have been proven to nurture beneficial gut bacteria. These foods provide prebiotics, essential substrates that stimulate the growth of healthy microbes. Conversely, diets high in processed foods and sugars can negatively impact microbiome diversity, leading to dysbiosis and an elevated risk of cancer. Recent research suggests that Mediterranean and plant-based diets can significantly improve gut health. These dietary strategies enhance microbial diversity and promote the presence of anti-inflammatory bacteria. By contrast, Western dietary patterns have been correlated with higher cancer incidence rates. Implementing dietary changes involves incorporating a variety of food groups which may be challenging for some aging individuals. However, gradual modifications toward healthier options can yield significant benefits. Personalized nutrition plans that consider individual preferences and lifestyles can be crafted to enhance adherence. As our understanding of the gut microbiome expands, nutritional interventions represent a promising frontier in cancer prevention among the aging population.
Understanding the gut-brain axis is vital when discussing the microbiome’s role in healthy aging and cancer prevention. This bidirectional communication pathway between the gut and brain underscores the systemic impact of gut health. Gut bacteria can influence brain function, impacting mood, cognition, and ultimately overall health. Chronic inflammation associated with dysbiosis may contribute to neurodegenerative disorders, hence indirectly affecting cancer progression. Studies have shown that improving gut health can enhance emotional well-being and reduce stress levels, both factors implicated in cancer risk. Mental health is increasingly recognized for its influence on physical health outcomes, highlighting the importance of holistic approaches in care for older adults. Incorporating lifestyle strategies such as mindfulness, exercise, and community engagement can support mental health and enhance gut microbiome diversity. Furthermore, interventions that promote a healthy microbiome may improve cognitive function, thereby fostering independent living in seniors. Such integrated approaches are essential for optimizing health outcomes in aging populations. By focusing on mental health alongside gut health, we can develop multifaceted strategies aimed at reducing cancer risks and enhancing quality of life for older adults.
Probiotics and Their Impact on Gut Health
Probiotics have gained popularity for their potential in supporting gut health and subsequently preventing cancer. These live microorganisms can restore microbial balance, especially in aging populations where dysbiosis is prevalent. Probiotic supplementation has been associated with improved immune response and reduced inflammation, both crucial factors in cancer prevention. Various strains of probiotics have shown potential in modulating gut microbiota effectively. For instance, Lactobacillus and Bifidobacterium species have been linked to reduced colon cancer risk. Additionally, certain probiotic products may enhance the efficacy of cancer treatments, such as immunotherapy. Ongoing research is exploring personalized probiotic formulations tailored for individual microbiome profiles. It is essential to note that probiotic efficacy can vary among individuals, necessitating a tailored approach. Older adults may benefit from specific probiotics suited to their health status and dietary habits. Moreover, ongoing consumer education on the importance of selecting high-quality, clinically-tested probiotics will empower individuals. As the field continues to evolve, integrating probiotics into dietary recommendations has the potential to significantly impact cancer prevention strategies for aging populations.
In conclusion, the gut microbiome plays a critical role in cancer prevention among aging populations. As we delve deeper into understanding the intricate relationships between gut health and overall well-being, various strategies emerge to mitigate cancer risk. Key practices include promoting dietary diversity, integrating probiotics, and fostering mental health approaches. It is essential for healthcare professionals to educate older adults about the importance of maintaining a healthy microbiome. The integration of nutritionists with oncologists and geriatric specialists can lead to comprehensive care strategies that emphasize prevention. Furthermore, community-based programs promoting healthy eating habits and physical activity can provide additional support. Research in this domain continues to evolve, paving the way for evidence-based interventions. As vitality in aging populations becomes a public health priority, understanding the microbiome’s implications becomes imperative. By fostering healthy gut ecosystems through dietary modifications and lifestyle improvements, we can enhance the quality of life and longevity. Ultimately, the goal is to mitigate cancer risk while promoting holistic wellness in older adults. This integrated approach holds great promise for improving health outcomes in our aging society.
Future Directions in Microbiome Research
Future research exploring the link between gut microbiome and cancer prevention must focus on several crucial areas. Investigating specific microbial profiles and their associations with cancer types may reveal targets for early intervention. Understanding the mechanisms through which gut microbes influence carcinogenesis is essential for developing preventive strategies. Furthermore, large-scale longitudinal studies are needed to assess the impact of dietary interventions on microbiome composition and cancer risk. Collaboration among multidisciplinary teams will enhance research quality, combining expertise from microbiology, oncology, and nutrition. Additionally, the role of the gut microbiome in shaping responses to various cancer therapies remains a ripe area for study. Identifying beneficial bacterial strains that could enhance treatment efficacy can significantly impact therapeutic outcomes. Finally, developing personalized strategies based on individual microbiome profiles presents a frontier in cancer prevention. As technology advances, metagenomics and bioinformatics will play pivotal roles in characterizing the microbiome’s complexity. Overall, continued investment in microbiome research is essential for unveiling the potential of gut health in combating cancer among aging populations. The future of personalized medicine could lead to targeted interventions that enhance longevity and quality of life.
In summary, the role of gut microbiome in aging populations is multifaceted and crucial for cancer prevention. As evidenced by ongoing research, maintaining gut health through diet, probiotics, and other lifestyle factors can significantly reduce cancer risks. The interplay between mental health and microbiome diversity emphasizes the need for a holistic approach in care. Encouraging healthier dietary practices and physical activity can enhance gut microbiota in older adults, supporting a healthy aging process. Importantly, a collaborative effort among healthcare providers can facilitate knowledge sharing, enabling personalized interventions tailored for individual needs. Enhancing public awareness about the gut microbiome’s influence on cancer and overall health will empower older adults to make informed lifestyle choices. As research advances, the integration of microbiome health into cancer prevention strategies will become increasingly vital. Furthermore, policymakers must prioritize funding for microbiome studies and interventions that target aging populations. Ultimately, prioritizing gut health will yield dividends in cancer prevention, leading to healthier aging and improved quality of life for the elderly. A proactive approach focusing on prevention through gut microbiome modulation will significantly impact future healthcare paradigms.