Gut Microbiome-Derived Metabolites and Their Influence on Liver Inflammation
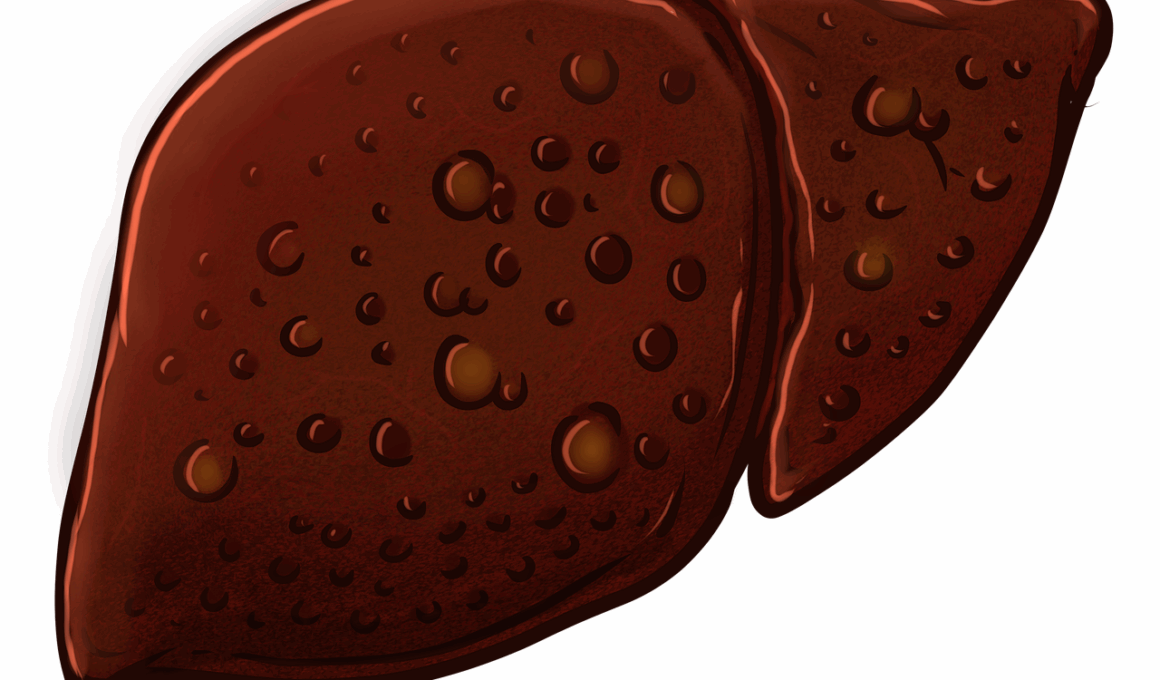
The gut microbiome plays a substantial role in overall health, particularly concerning liver function and inflammation. The relationship between gut bacteria and the liver, known as the gut-liver axis, reveals how microbiome-derived metabolites impact hepatic processes. These metabolites include short-chain fatty acids (SCFAs), bile acids, and various signaling molecules. SCFAs are produced by the fermentation of dietary fibers and exert anti-inflammatory effects on liver tissues. Following diet and lifestyle changes, alterations in bacterial composition may lead to variations in metabolite production. Long-term imbalances can result in liver disease or worsening inflammation. Understanding this connection emphasizes the importance of maintaining a healthy gut microbiome for optimal liver health. Adopting dietary practices that promote beneficial gut bacteria can therefore be key in preventing liver inflammation and diseases. Equally important, identified metabolites may serve as potential biomarkers for liver conditions. This relationship not only enhances our comprehension of liver inflammation but may also offer novel therapeutic targets and strategies for remedying various hepatic diseases, benefiting individuals suffering from liver-related complications.
The Role of SCFAs in Liver Health
Short-chain fatty acids, predominantly butyrate, propionate, and acetate, are important metabolites of gut microbiota. They provide energy for colon cells, maintaining gut barrier integrity, which can influence liver health. SCFAs are absorbed into the bloodstream after dietary fiber fermentation. Their beneficial impact includes regulating immune responses and acting as anti-inflammatory agents. The modulation of gut bacteria through a fiber-rich diet allows enhanced SCFA production and subsequently decreases hepatic inflammation. Additionally, SCFAs interact with various signaling pathways, possessing the ability to stimulate GLP-1 secretion. By promoting insulin sensitivity, SCFAs help in maintaining metabolic homeostasis, consequently reducing the risk of fatty liver disease. The therapeutic potential of SCFAs lies in their capacity to mitigate non-alcoholic fatty liver disease, characterized by excessive fat accumulation in hepatocytes. This interplay signifies the necessity for dietary fibers that foster healthy gut microbiota, leading to improved liver health. Various studies are converging on SCFAs as promising targets for dietary interventions aimed at liver protection. Diet modifications should focus on fiber intake to harness the full potential of SCFAs for liver health and inflammation management.
In addition to SCFAs, bile acids are vital microbiome-derived metabolites that impact liver inflammation. Bile acids are synthesized in the liver and form part of the gut microbiome interaction. Bile salts undergo transformation by gut bacteria, which converts primary bile acids into secondary bile acids. These transformations are essential for metabolic regulation and maintaining gut homeostasis. Secondary bile acids have been observed to exert anti-inflammatory effects and can help regulate bacterial composition within the gut. Alterations in bile acid profiles can herald inflammation and fat deposition in the liver, contributing to liver diseases such as cholestatic liver disease. Bile acid signaling via the farnesoid X receptor (FXR) and TGR5 receptors is crucial for liver inflammation modulation. The investigation of bile acids as metabolic mediators provides valuable insights into their therapeutic potential. As emerging evidence suggests that modulating the gut microbiome can alter bile acid profiles, interventions that promote beneficial gut flora may alleviate liver inflammation. Exploring the dynamics of microbiome interactions underscores the importance of the gut-liver axis as a therapeutic frontier.
Furthermore, specific dietary patterns can dramatically influence the gut microbiome and thereby affect liver health. Diets high in saturated fats and sugars have been shown to disturb gut microbiota diversity, leading to increased inflammation. Conversely, a balanced diet characterized by fruits, vegetables, whole grains, and healthy fats fosters a beneficial microbiome. Probiotic and prebiotic foods can enhance microbial diversity. Fermented foods such as yogurt or sauerkraut supply live beneficial bacteria, contributing positively to gut health. Moreover, prebiotics, including inulin and oligosaccharides, serve as food sources for beneficial bacteria, promoting their growth within the gut. The shift in microbial populations can enhance SCFA and bile acid production, mitigating liver inflammation. Lifestyle modifications such as reducing alcohol intake and regular physical activity can further reinforce these dietary changes. Ultimately, integrating healthy dietary practices may establish a sustainable environment for beneficial gut bacteria, promoting liver health while reducing systemic inflammation. The intersection between diet and gut microbiota stresses the need for considered lifestyle choices to nourish both gut and liver health.
Emerging research is emphasizing the potential of microbiome-derived metabolites as biomarkers for liver disease. With the intricate relationship between gut health and liver inflammation, identifying specific bacterial metabolites could lead to innovative diagnostic approaches. Several studies have linked altered metabolite profiles to liver conditions, setting a precedent for their use in clinical settings. Parameters such as SCFAs and bile acids are particularly promising due to their direct ties to inflammation pathways. Monitoring these metabolites could provide insights into disease progression or therapeutic efficacy. Additionally, microbiome composition analysis via stool samples may offer non-invasive methods to assess liver health. However, further research is critical to validate these biomarkers across diverse populations and disease states. Clinical trials investigating microbiota modulation approaches may also enhance our understanding of bacteriotherapy’s role in liver diseases. Given the rising incidence of liver disorders, the integration of microbiome-focused diagnostics presents a compelling avenue for early intervention. As we advance in characterizing gut metabolites, opportunities to personalize treatment protocols for liver ailments arise, paving the way for innovative and patient-centered health solutions.
Microbiome and Hepatic Disease Relationship
The interconnection between the gut microbiome and hepatic diseases extends beyond inflammation to various liver disorders, including cirrhosis and hepatocellular carcinoma. Evidence indicates a bidirectional relationship: liver diseases can influence gut bacteria, and dysbiosis can precipitate liver pathologies. Chronic liver diseases, such as cirrhosis, may lead to increased intestinal permeability, permitting translocation of bacteria and their products into the bloodstream. This translocation fuels systemic inflammation and further exacerbates liver damage. Conversely, certain gut microbiota alterations can predispose individuals to hepatic disorders. For instance, a diminished presence of beneficial bacterial species may enhance susceptibility to liver conditions. Understanding these reciprocal effects allows for targeted preventative strategies in at-risk populations. Research suggests that restoring microbial balance may alleviate liver inflammation, presenting microbiota as therapeutic targets. Trials examining probiotic or prebiotic therapies in individuals with hepatic impairment showcase promising results. The role of the gut microbiome in influencing hepatic pathophysiology signifies the emerging frontier of microbiota-centered interventions for treating liver diseases.
Finally, the exploration of the therapeutic potential of gut microbiome modulation opens exciting avenues for improving liver health. The rational design of probiotics and prebiotics shows promise in maintaining a healthy microbial balance while mitigating liver inflammation. Formulations can be tailored based on an individual’s unique gut ecology, seeking to restore diversity lost to poor diet or health conditions. Recent trials focusing on targeted probiotic strains highlight their efficacy in reducing hepatic inflammatory markers, showcasing their ability to influence metabolic pathways beneficially. Furthermore, lifestyle recommendations encompassing dietary habits and physical activity can synergize with microbiome modulation efforts. Personalized nutrition, targeting specific microbial populations, may amplify the benefits realized through probiotics or dietary changes. As research evolves, treatments based on gut microbiome optimization can pave the way for enhanced liver health. Prevention may become more effective as we uncover the intricacies between gut bacteria and hepatic disease. Ultimately, the gut-liver axis underscores the importance of microbiome stewardship in fostering health and preventing disease.