Antibiotic Use and Changes in Gut Microbial Gene Expression
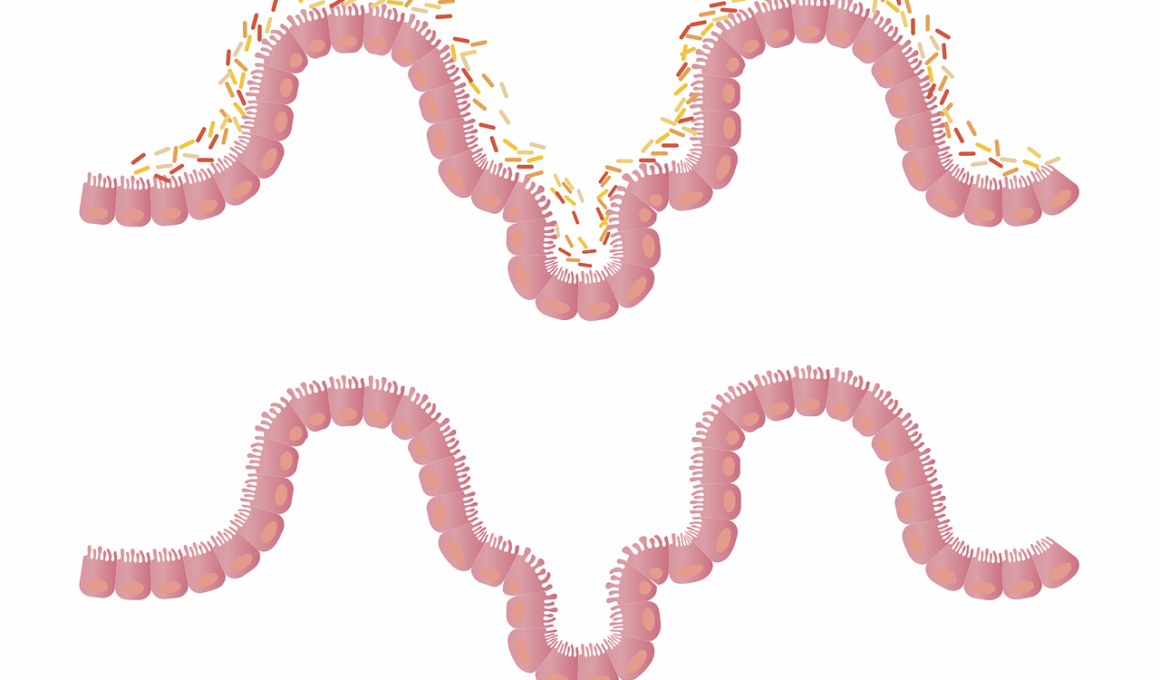
Antibiotics are compounds used to treat bacterial infections, but their impact on the gut microbiome can be profound. The gut microbiome consists of trillions of microorganisms, including bacteria, fungi, and viruses, and is integral to gut health and overall wellbeing. When antibiotics are administered, they do not selectively target harmful bacteria. Instead, they often disrupt the balance of beneficial microorganisms, which may lead to a shift in microbial diversity. This alteration can influence various bodily functions, including digestion and immune response. Studies indicate that both short-term and long-term antibiotic use can impact gut microbial gene expression significantly. Gene expression refers to the active processes that result in the synthesis of proteins, which can affect metabolism and health outcomes. For instance, certain beneficial bacteria may decrease in numbers due to antibiotic exposure, while opportunistic pathogens might proliferate. Understanding these dynamics is crucial for developing strategies to mitigate the adverse effects of antibiotics on the gut microbiome. In this article, we will explore how antibiotics influence gut microbial gene expression and the potential long-term consequences on human health.
The implications for gut health are profound when antibiotics alter microbial gene expression. Gut microorganisms perform essential functions that contribute to overall health. For instance, they aid in the digestion of complex carbohydrates, synthesize essential vitamins, and protect against pathogens. Disruption of their balance can lead to gastrointestinal symptoms such as diarrhea, bloating, and abdominal pain. A well-functioning microbiome is crucial for maintaining an intact gut barrier, preventing the translocation of harmful bacteria and toxins into the bloodstream. Research has shown that antibiotic-induced dysbiosis can persist long after the antibiotic course has ended. This persistent state can lead to an increase in inflammatory responses and vulnerability to infections. Furthermore, the reduced diversity of gut microbiota can limit the resilience of the microbial community, making recovery difficult. A healthy microbiome is key to developing tolerance against pathogenic organisms while enhancing the efficacy of vaccines. Therefore, understanding how antibiotics reshape microbial gene expression can inform prescribing practices and lead to the development of strategies aimed at preserving gut health during and after antibiotic therapy.
The timing and type of antibiotic therapy play significant roles in determining the extent of changes in microbial gene expression. Broad-spectrum antibiotics, while effective against a wide range of bacteria, can cause more significant disruptions compared to narrow-spectrum antibiotics, which target specific pathogens. Research suggests that certain classes of antibiotics can lead to rapid and substantial changes in the gut microbiome, altering gene expression profiles associated with metabolism and immune modulation. For example, studies demonstrate that antibiotics can downregulate genes involved in the synthesis of short-chain fatty acids, essential metabolites that help regulate inflammation and maintain gut health. The timing of administration is also pivotal; early-life antibiotic exposure can have long-lasting effects on the developing gut microbiome of infants, potentially predisposing them to conditions such as obesity or allergies later in life. Moreover, the repeated cycles of antibiotic treatment can result in a more entrenched shift in microbial populations and gene expression patterns. Research into these phenomena can offer critical insights into mitigating antibiotic resistance and fostering the recovery of a healthy gut microbiome.
Strategies for Protecting Gut Health During Antibiotic Use
To preserve gut microbiome health during antibiotic treatment, several strategies can be employed. Probiotic supplementation is one option that may help restore balance in gut flora after antibiotic courses. Probiotics are live microorganisms that confer health benefits when administered in adequate amounts. Individuals taking antibiotics can benefit from taking probiotics alongside their medication, thereby lessening the disruptive effects on microbial diversity. In addition, consuming prebiotic-rich foods can promote the growth of beneficial bacterial populations in the gut. Prebiotics are non-digestible fibers found in foods such as garlic, onions, and bananas. These substances serve as food for beneficial gut bacteria, assisting in their recovery during and after antibiotic use. Furthermore, maintaining a balanced diet rich in fruits, vegetables, and whole grains can support gut health by providing essential nutrients and promoting diverse microbial populations. Hydration is also critical; adequate fluid intake supports mucosal health and helps manage gastrointestinal side effects that may arise during antibiotic therapy. Awareness of these strategies can significantly improve the outcomes for individuals undergoing antibiotic treatment.
Research indicates that the consequences of antibiotic usage on the gut microbiome extend beyond immediate health effects. Long-term changes in microbial gene expression may be tied to chronic diseases. For example, evidence has linked shifts in gut microbiota composition caused by antibiotics to the development of metabolic disorders such as obesity and type 2 diabetes. Furthermore, recent studies suggest that alterations in the gut microbiome might contribute to conditions like inflammatory bowel disease and allergies. The mechanisms by which antibiotics impact microbial populations and gene expression are intricate, often involving interactions between host immunity and microbial activity. For instance, immune responses generated by certain gut bacteria can influence how the body processes nutrients and manages inflammation. Consequently, understanding these connections can lead to more targeted therapeutic interventions and preventative measures against chronic diseases. The ongoing research in this field underscores the importance of viewing antibiotics not merely as tools for treating infections but also as agents that can profoundly influence gut microbiome architecture and function over time.
The relationship between antibiotics and gut microbial gene expression also raises questions about the future of antimicrobial therapies. With the rising concerns around antibiotic resistance, the need for alternative treatments and complementary approaches is becoming increasingly evident. Novel therapies, such as bacteriophage therapy and antimicrobial peptides, aim to combat specific pathogens while preserving beneficial microbiota. Meanwhile, more precise and judicious antibiotic use is promoted in clinical settings. Guidelines encouraging the limited use of broad-spectrum antibiotics emphasize the importance of targeted approaches based on microbiome assessments. Additionally, advances in therapeutic stool transplantation could offer a solution for restoring gut microbiota after a course of antibiotics. These novel treatment strategies not only focus on eradicating pathogens but also prioritize maintaining microbial diversity and promoting overall gut health. As research continues to evolve, the development of microbiome-targeted therapies and a better understanding of antibiotics’ impacts signal a paradigm shift in antimicrobial stewardship—steering focus towards holistic health rather than merely treatment of microbial infections.
Conclusion and Future Perspectives
In summary, the impact of antibiotics on gut microbial gene expression is essential for grasping their broader implications on human health. The disruption caused by antibiotic therapy can lead to significant changes in microbial community structure and functionality, affecting digestion, immunity, and even chronic disease development. Recognizing the importance of a balanced gut microbiome highlights the need for cautious antibiotic use and the incorporation of strategies that support microbial health during treatment. Future research must focus on elucidating the mechanisms behind microbial interactions, gene expression alterations, and the link to health outcomes. Furthermore, as precision medicine evolves, tailoring antibiotic therapies based on an individual’s gut microbiome profile could pave the way for more effective and safer treatment strategies. The shift towards integrating gut health into prescribing practices will not only safeguard against the adverse effects of antibiotics but also enhance overall healthcare outcomes. By continuing to explore the complexities of the gut microbiome and antibiotic interactions, we can move towards a future where health is optimized and maintained through informed medical practices and enhanced understanding of microbial ecosystems.
Ultimately, the ongoing dialogue surrounding antibiotics and the gut microbiome must include healthcare professionals, researchers, and patients alike. Engaging patients in conversations about the impact of antibiotics on gut health can promote shared decision-making and informed choices regarding their treatment. Encouraging discussions around the benefits and risks of antibiotic use empowers patients to advocate for their health and make choices that best suit their individual circumstances. Educational initiatives targeting both healthcare providers and the public can cultivate a better understanding of the gut microbiome’s role in health and disease. Adopting a public health perspective is essential in addressing the challenges posed by antibiotic resistance and gut microbiome dysbiosis. As research progresses, collaboration across scientific disciplines will be crucial to uncovering innovative solutions to preserve and restore gut health. The legacy of antibiotic therapy must be continuously reassessed in light of emerging evidence to ensure patient safety, promote health at the community level, and safeguard the microbiomes of future generations. With a commitment to responsible antimicrobial stewardship and ongoing research, we can strike a balance that prioritizes effective treatment while preserving the integrity of our vital gut ecosystems.