Is There a Link Between Gut Microbiome and Eosinophilic Esophagitis?
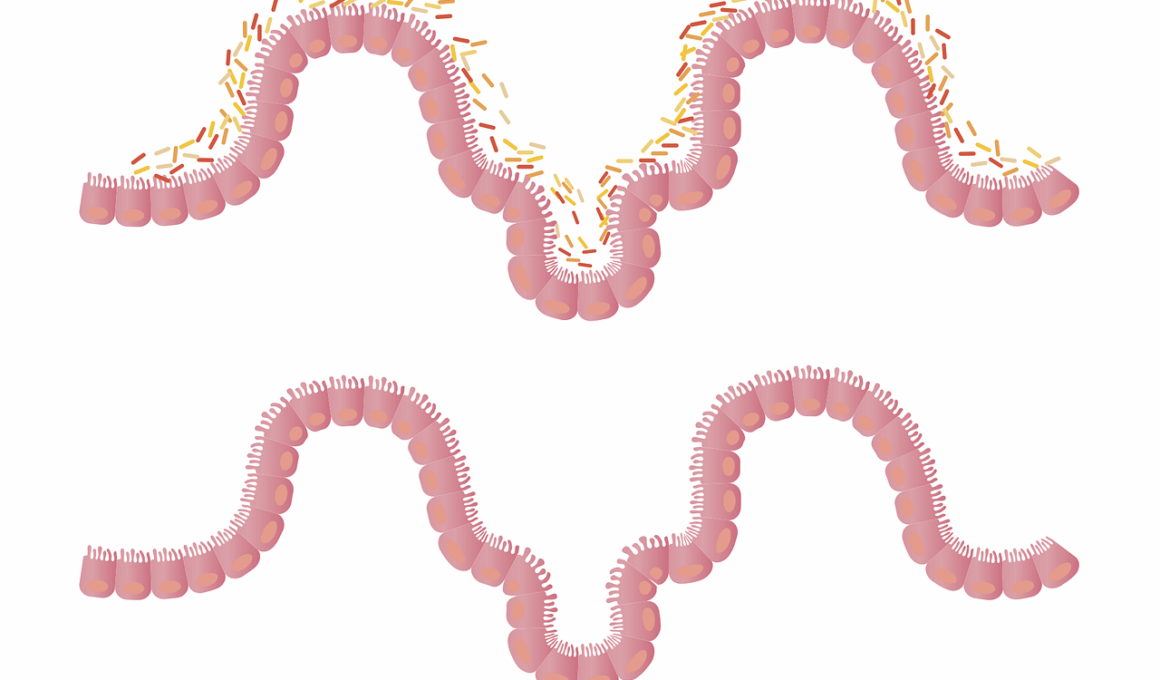
The connection between gut microbiome and eosinophilic esophagitis (EoE) is an increasingly researched topic, highlighting how certain food allergies may influence gut health. EoE is a chronic inflammatory condition affecting the esophagus, often triggered by food allergies, particularly in children. The gut microbiome, comprising trillions of microorganisms, plays a crucial role in digestive health, immune system regulation, and inflammation control. When the microbiota is imbalanced, it can lead to adverse effects on gut integrity, promoting conditions like EoE. Understanding this link is essential for developing effective treatment strategies. By investigating how microbiome composition interacts with food allergies, researchers aim to identify specific bacterial profiles associated with EoE. This research could pave the way for personalized dietary interventions, focusing on restoring healthy gut flora, thereby alleviating symptoms. EoE management often involves dietary elimination, but incorporating insights from gut microbiome studies may enhance efficacy. Continued research in this area can also contribute greatly to food allergy management, enhancing overall digestive health. In conclusion, the relationship between gut microbiome and EoE is complex but presents promising avenues for exploration.
In examining the gut microbiome’s role, studies suggest that certain bacteria may influence allergic responses and inflammation levels within the esophagus. For individuals with EoE, alterations in the diversity and composition of gut flora could mediate the immune response to specific food allergens, leading to esophageal inflammation. Some research indicates that individuals with EoE often have lower microbial diversity than healthy individuals, suggesting a potential imbalance contributing to the allergic condition. A hyperactive immune system may react aggressively to innocuous dietary components if beneficial gut bacteria are lacking. Furthermore, gut bacteria like Lactobacillus and Bifidobacterium can help modulate immune responses, potentially protecting against food allergies. Probiotic therapies, which introduce beneficial bacteria into the gut, are being explored as a treatment option for EoE. These approaches could help restore microbial balance and reduce inflammation. However, further clinical trials are needed to understand the long-term effects and efficacy of probiotics in managing EoE. Effective dietary management and understanding the gut microbiome’s influence on the condition may ultimately enhance patient care and improve quality of life for those affected.
Nutrition plays a critical role in the health of the gut microbiome, impacting conditions like eosinophilic esophagitis. A diet rich in whole foods, fiber, and fermented products is beneficial for maintaining a balanced microbiome. Conversely, processed foods, high sugar, and fats can lead to dysbiosis, exacerbating EoE symptoms. Identifying food triggers is essential for individuals diagnosed with EoE, as removing certain allergens from their diet can alleviate symptoms. Common triggers include dairy, wheat, soy, peanuts, tree nuts, fish, and eggs. Additionally, this process requires patience and careful monitoring to achieve the best results. Scientific evidence suggests that integrating diverse plant-based foods may bolster microbial diversity, providing a protective effect against allergies while ensuring essential nutrients. Medical professionals often recommend guidance from a nutritionist to create a comprehensive dietary plan tailored to enhancing gut health. Moreover, understanding the importance of balanced nutrition helps empower patients to make informed choices, subsequently improving their overall well-being. As research into the gut microbiome continues, it emphasizes the need for a holistic approach to EoE management that encompasses both dietary choices and lifestyle alterations.
The Role of Prebiotics in Gut Health
Prebiotics are non-digestible food components that promote the growth and activity of beneficial gut bacteria. They differ from probiotics, which are live microorganisms. Prebiotics, found in foods such as garlic, onions, asparagus, and bananas, serve as food sources for healthy gut bacteria. Increasing the intake of prebiotics can play a crucial role in managing EoE by enhancing the gut microbiome’s overall diversity and encouraging the growth of beneficial bacteria. This increased diversity may help mitigate abnormal immune responses associated with food allergies. Research highlights that a flourishing gut microbiome can better regulate inflammation and immune activity, potentially reducing the severity of EoE symptoms. Prebiotics may improve gut barrier function and reduce intestinal permeability, further helping to minimize allergy triggers. It is vital for EoE patients to work towards balancing gut health through a diet rich in prebiotics. Consulting with healthcare professionals can guide individuals in incorporating various prebiotic-rich foods into their diets effectively. This dietary strategy is likely to support a healthier digestive environment, allowing EoE patients to tolerate potential allergens more appropriately and comfortably.
The use of specific diets, such as the elimination diet, remains a cornerstone in managing eosinophilic esophagitis. This dietary approach requires patients to remove identified allergens and then slowly reintroduce them while monitoring symptoms. Understanding the role of the gut microbiome can enhance this process, making it vital for patients to engage effectively with their healthcare providers. By focusing on individual responses during dietary reintroductions, those affected by EoE can determine their tolerance levels more accurately. Furthermore, exploring the link between certain dietary constituents and gut microbiome alterations can provide deeper insights into managing EoE symptoms. Dietitians and allergists can work together to develop personalized plans that consider both dietary needs and the intricacies of gut health. Ultimately, tracking how the gut microbiome evolves with dietary changes will be critical for optimizing long-term management strategies. This collaborative approach can lead to improved patient outcomes, with a comprehensive understanding of how dietary choices impact both gut microbiome and EoE. Regular follow-ups and adjustments to dietary plans based on microbiome health could significantly enhance quality of life for those managing the condition.
Current research trends are increasingly focusing on the interplay between gut microbiome, food allergies, and eosinophilic esophagitis. Promising studies show that modulation of the gut microbiome through dietary interventions may yield significant improvements in patients’ inflammatory symptoms. Ongoing clinical trials are evaluating the impact of various probiotics, prebiotics, and dietary patterns on EoE management and overall gut health. Researchers aim to determine specific microbial strains or combinations that may provide lasting benefits to EoE patients. Furthermore, understanding the genetic factors influencing individual responses to food allergens is essential, paving the way for tailored interventions. The potential for novel therapeutics targeting gut microbiome modulation is exciting, offering hope for enhanced treatment protocols in the future. Integrating findings from these studies into clinical practice could revolutionize EoE management. By advocating for further investigations into this complex relationship, healthcare professionals can remain at the forefront of approaches to manage EoE efficiently. Patient education on dietary impacts and microbiome health remains crucial, ensuring that individuals are empowered to make informed decisions regarding their dietary choices.
While much progress has been made in understanding the connections between the gut microbiome and eosinophilic esophagitis, more research is necessary. Longitudinal studies tracking microbiome changes and associated EoE symptoms will provide essential information to optimize treatment options. Researchers are also encouraged to explore the environmental factors contributing to gut dysbiosis, as lifestyle influences could enhance overall health outcomes. Such comprehensive research will not only strengthen the connection between diet, gut health, and EoE but also emphasize the importance of personalized medical approaches. Engaging patients in discussions about their dietary habits and lifestyle can yield rich qualitative data, shedding light on the unique interactions between gut health and eosinophilic esophagitis. As understanding improves, so does the potential for breakthroughs in treatment options, including probiotics and dietary counseling. The future of EoE management may hinge on harnessing microbiome research to offer patients more effective and targeted therapies. Integrative approaches that incorporate the patient’s microbiome profile may provide pathways toward successfully controlling symptoms and achieving lasting relief.
In conclusion, the intricate relationship between the gut microbiome and eosinophilic esophagitis highlights the need for further research and tailored interventions. Understanding these connections can significantly enhance disease management strategies for patients struggling with this condition. By integrating dietary modifications, exploring useful probiotics, and understanding the significance of gut health, individuals can advocate for improved outcomes. It is essential to consider collaborative healthcare approaches that include specialists in nutrition, immunology, and gastroenterology. A holistic treatment plan not only addresses physical symptoms but also empowers individuals through education and support. As science advances, patients can anticipate more comprehensive management options based on the latest findings. The ongoing exploration of gut-microbiome relations provides hope for those suffering from eosinophilic esophagitis. By embracing these evolving insights, the path to symptom relief becomes clearer. Thus, the future of management in EoE may be brighter, with integration of gut microbiome knowledge offering improved quality of life for many. Together, through continued research and shared knowledge, a clearer understanding of the link between gut health and eosinophilic esophagitis will emerge.