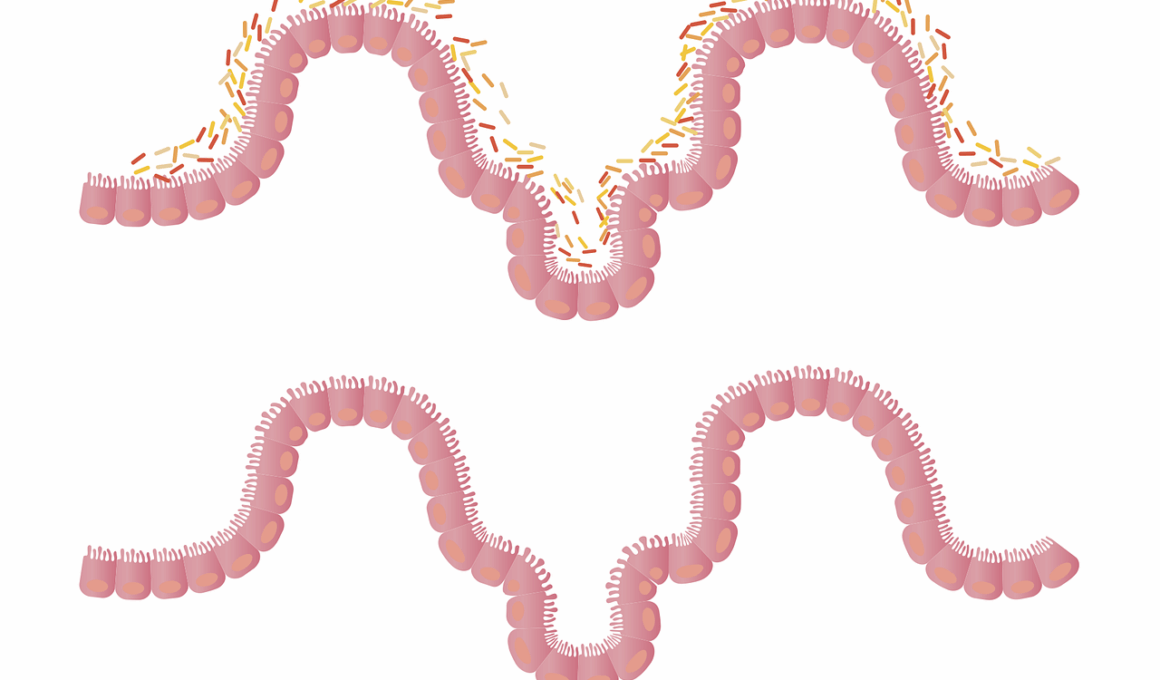
The Role of Antibiotics in Disrupting Gut Microbes
The gut microbiome is a vast ecosystem comprised of trillions of microorganisms, including bacteria, fungi, and viruses that thrive in our digestive tract. It plays a crucial role in our overall health, influencing everything from digestion to immune system function. However, the use of antibiotics can significantly disrupt this delicate balance, leading to various health complications. Antibiotics are designed to eliminate harmful bacteria; however, they are indiscriminate and target both good and bad bacteria in the gut. This can lead to a decrease in microbial diversity, which is vital for a healthy gut. The reduction in diversity can make the gut more susceptible to infections and other diseases. Furthermore, this imbalance can result in digestive issues, such as diarrhea and bloating, and may even have long-term impacts on mental health by influencing the gut-brain axis. Understanding the implications of antibiotic use on the microbiome is essential for making informed decisions about the use of these medications. Inappropriate use can lead to antibiotic resistance, posing a larger risk to public health and well-being.
Antibiotics disrupt gut microbiota composition, leading to a range of negative health outcomes. One significant effect is the overgrowth of harmful pathogens. Upon antibiotic treatment, beneficial bacteria are often wiped out, creating an opportunity for harmful pathogens like Clostridium difficile to flourish. This can lead to severe infections that are difficult to treat. Additionally, the loss of beneficial microbes results in compromised gut barrier function, which may lead to increased intestinal permeability, commonly referred to as “leaky gut.” In this condition, toxins and bacteria can enter the bloodstream, triggering inflammation and contributing to various autoimmune conditions. Research indicates that this disruption can have lasting effects, sometimes requiring months or years for the gut microbiota to recover fully. Incorporating probiotics and fermented foods during and after antibiotic treatment can aid in restoring microbiome balance. However, it’s essential to consult healthcare professionals about safe practices. Overall, recognizing the significant repercussions of antibiotic use is crucial to mitigate its negative effects on a well-balanced gut microbiome. Responsible utilization of antibiotics, combined with dietary considerations, can help maintain gut health.
The Importance of Gut Microbiome Diversity
The diversity of the gut microbiome is a critical factor in maintaining overall health. A well-fed microbiome is crucial for optimizing nutrient absorption, synthesizing vitamins, and supporting immune function. When antibiotics are prescribed, they can drastically reduce this vital microbial diversity. The loss of beneficial microbial communities not only promotes the growth of harmful bacteria but can also lead to dysbiosis, a condition characterized by an imbalance in the gut microbiota. Increased dysbiosis is linked to numerous health concerns, including obesity, diabetes, and inflammatory bowel disease. Studies suggest that a diverse gut microbiome can also enhance metabolic flexibility and resilience against external stressors. To promote microbiome diversity, individuals should consume a varied diet rich in fruits, vegetables, whole grains, and fermented products. These foods provide essential nutrients and promote the growth of a wide range of beneficial microorganisms. Moreover, regular physical activity and stress management can significantly influence gut health. It is essential to recognize that preserving gut microbiome diversity is not only beneficial for digestive health but also foundational for enhancing overall wellness.
The impact of antibiotics on the gut microbiome extends beyond the gastrointestinal tract; it can manifest in various systemic health issues. Disruption of the gut microbiome has been linked to mood disorders, such as depression and anxiety. This connection is primarily due to the gut-brain axis, a pathway where gut microbes communicate with the brain. Imbalances caused by antibiotic use can produce neuroactive compounds that affect mood regulation. Therefore, individuals on antibiotics may experience increased symptoms of anxiety and depression. In addition, studies have explored the connection between antibiotic-induced dysbiosis and the development of metabolic disorders, such as obesity and type 2 diabetes. The microbiome influences the body’s metabolism, energy harvest, and storage, thus playing a role in weight management. Recognizing these implications can assist healthcare providers in offering a more holistic approach to treatment regimens that include alternatives to antibiotics, thereby supporting gut health proactively. As awareness about the impacts grows, it becomes imperative for patients to discuss microbiome considerations with their healthcare providers and adopt personalized health strategies.
Restoring Gut Health after Antibiotic Use
Restoring gut health following antibiotic treatment is essential for achieving long-term wellness. After completing a course of antibiotics, it’s advisable to implement dietary strategies that promote the re-establishment of beneficial gut bacteria. Incorporating probiotic-rich foods, such as yogurt, kefir, and sauerkraut, can facilitate the recovery process. These foods contain live beneficial bacteria that help replenish the microbiome, restoring its balance. Additionally, prebiotics, which are non-digestible fibers found in foods like garlic, onions, and asparagus, can nourish and stimulate the growth of beneficial microbes. Staying well-hydrated is equally important; adequate water intake supports the digestive system and contributes to the efficient elimination of toxins from the body. Furthermore, engaging in regular physical activity enhances overall gut health by promoting circulation and supporting a balanced microbiome. Limiting sugar and processed foods, which can feed harmful bacteria and exacerbate dysbiosis, is also advisable. Establishing a healthy post-antibiotic regimen can mitigate the adverse effects of these medications, leading to improved gut health and overall well-being.
The rise of antibiotic resistance underscores the importance of responsible antibiotic use. With the growing prevalence of resistant strains of bacteria, healthcare professionals must exercise caution in prescribing antibiotics. Encouraging patients to adhere to prescribed dosages and durations is vital in preserving the efficacy of these medications. Additionally, antimicrobial stewardship programs within healthcare settings can promote the judicious use of antibiotics. Patients can contribute by engaging in open conversations with healthcare providers to explore alternative treatment options, such as herbal remedies or homeopathic treatments. Preventing infections through proper hygiene, vaccination, and lifestyle changes can significantly reduce the need for antibiotics. Moreover, enhancing public awareness about the consequences of inappropriate antibiotic use is fundamental in combating resistance. Ultimately, this collective effort requires collaboration between healthcare providers, patients, and policymakers. Only by prioritizing responsible antibiotic prescribing can we protect not just individual gut health but also the microbial ecosystems essential for maintaining global health.
Future Directions for Gut Microbiome Research
The field of gut microbiome research is rapidly evolving, presenting numerous opportunities for future study. Investigating the complex interactions between dietary habits, lifestyle, and antibiotic use will prove invaluable in understanding their cumulative impacts on gut health. Personalized microbiome analysis, assessing an individual’s specific gut flora, holds promise for tailoring dietary recommendations and treatment plans. Future research could also explore the potential of targeted probiotics, designed to restore specific imbalances caused by antibiotic use. Leveraging advanced microbiome profiling techniques can yield more precise data on microbial diversity and activity. Additionally, interdisciplinary approaches that incorporate insights from nutrition, genetics, and immunology could lead to a holistic understanding of gut health. Another promising area is the development of new antibiotics that selectively target harmful bacteria without disrupting beneficial ones, potentially reducing the incidence of dysbiosis. This continued exploration into the gut microbiome’s role in overall health and disease management highlights the necessity of prioritizing these investigations. Such efforts have the potential to transform health strategies and guide future clinical practices.
In conclusion, the role of antibiotics in disrupting gut microbes cannot be overstated. Understanding their impact is essential for maintaining a healthy gut microbiome. Balancing effective antibiotic use while safeguarding microbial diversity should be a priority for both patients and healthcare providers alike. By emphasizing responsible use, coupled with dietary adjustments and lifestyle changes, restoration of gut health is achievable. Continued research and education on this topic remain vital as we strive for improved understanding and better health outcomes for individuals worldwide. As the science of the gut expands, we become better equipped to support microbial ecosystems and enhance wellness in the face of antibiotic challenges.