The Bidirectional Interaction Between Drugs and Gut Microbiome
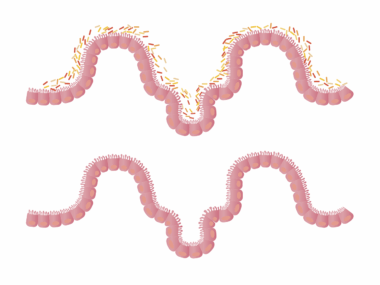
The gut microbiome is a complex ecosystem, crucial for digestion, immune function, and overall health. Recent research has highlighted how certain medications can significantly impact this microbial balance. Disruption in the microbiome can lead to a variety of health issues, including digestive disorders, obesity, and autoimmune conditions. This dynamic interaction means that not only can drugs alter gut bacteria, but the state of the gut microbiome can also influence a person’s response to medications. Understanding these interactions is essential for patients and healthcare providers to optimize treatment plans. Many medications, particularly antibiotics, are known to decrease microbial diversity, which is a key aspect of gut health. This can lead to potential side effects, such as increased susceptibility to infections. It is also suggested that the gut microbiome plays a role in drug metabolism, potentially leading to variations in drug efficacy among individuals. Therefore, evaluating the effect of medications on gut health could pave the way for more personalized medicine, maximizing therapeutic benefits while minimizing adverse reactions. Continued research in this field is vital to develop strategies that preserve a healthy gut microbiome during pharmacological treatments.
Antibiotics and Their Impact on Microbial Diversity
Antibiotics are often vital for treating bacterial infections, but they can disrupt the gut microbiome’s delicate equilibrium. Use of antibiotics can lead to a decrease in microbial diversity, a phenomenon that has been well documented in recent studies. Reduced diversity in gut flora may contribute to various complications, including Clostridium difficile infections, which can be life-threatening. The extent and duration of antibiotic use significantly dictate the degree of harm to gut microbiota. Studies show that even a short course of broad-spectrum antibiotics can provoke a substantial shift in microbial composition. After antibiotic treatment, it may take weeks or even months for the gut microbiome to return to its pre-treatment state. In some cases, certain bacterial populations may not fully recover, creating a long-term imbalance. Furthermore, research indicates that such disruptions could also affect drug efficacy, highlighting the need for cautious prescribing practices. Patients should be adequately informed about these risks, and healthcare providers may consider alternatives like probiotics to help mitigate adverse effects. Probiotics might play a significant role in maintaining or restoring gut health post-antibiotic therapy, thereby improving overall treatment outcomes.
Particularly, nonsteroidal anti-inflammatory drugs (NSAIDs) have also shown detrimental effects on gut microbiota. These medications are commonly prescribed for pain and inflammation issues, but studies reveal their potential to alter gut microbial communities and reduce diversity similarly to antibiotics. While they provide significant relief for painful conditions, NSAIDs can lead to gastrointestinal side effects like ulcers or bleeding. The relationship between NSAIDs and gut health necessitates careful consideration, especially for long-term users. Research has demonstrated that the consistent intake of NSAIDs can impair intestinal permeability and stimulate dysbiosis. This alteration in gut flora could not only limit drug absorption but also exacerbate underlying gut issues. It may result in a cascade of unintended effects that complicate treatment plans. Advocating for a well-balanced diet rich in fiber may help mitigate some NSAID-related gastrointestinal disturbances. Incorporating probiotics alongside NSAIDs during treatment may help preserve microbial diversity, promoting a healthier gut. Thus, understanding patient history and discussing dietary influences can play a crucial role in mitigating the adverse effects of NSAIDs on gut microbiome, therefore enhancing the effectiveness of these medications.
Antidepressants and Microbiome Interactions
Antidepressants represent another class of medications with significant implications for gut microbiome interactions. The relationship between mental health and gut health, often referred to as the gut-brain axis, emphasizes the impact of gut bacteria on mood and behavior. Several studies suggest that selective serotonin reuptake inhibitors (SSRIs), a common type of antidepressant, can influence the gut microbiome composition. Changes in microbial communities may not only modify drug metabolism but also contribute to the overall therapeutic response of these medications. Patients on SSRIs may experience gastrointestinal side effects related to shifts in gut bacteria, leading to symptoms such as nausea, diarrhea, or constipation. Thus, the interplay between antidepressant therapy and gut microbiome diversity is essential for optimizing treatment outcomes. Additionally, some research indicates that enhancing gut microbiota through dietary changes may serve as a complementary approach to traditional antidepressant treatments. Probiotics and fermented foods rich in beneficial bacteria might help improve symptoms of depression by supporting gut health. Therefore, healthcare providers should consider these interactions when prescribing antidepressants, ensuring a holistic approach to mental health management.
Recent studies have investigated the role of proton pump inhibitors (PPIs) in influencing gut microbiome stability. PPIs are widely utilized to manage acid reflux and peptic ulcers by inhibiting stomach acid production. However, emerging evidence suggests that long-term PPI usage may negatively impact the gut microbiome. Reduced gastric acidity can lead to alterations in microbial composition, potentially increasing susceptibility to gastrointestinal infections. The implications of this microbiome modulation can affect overall health, considering the gut’s role in immune function. Dysbiosis, resulting from prolonged PPI use, could also exacerbate digestive disorders and negatively influence drug metabolism. For example, altered gut flora can lead to variable responses to medications such as anticoagulants, affecting their therapeutic efficacy. Recent findings advocate for more cautious prescribing practices concerning PPIs, emphasizing the importance of considering gut health. Providers should evaluate the risk-benefit ratio for patients requiring long-term acid suppression therapy. Moreover, simple lifestyle changes and dietary modifications can serve as adjuncts to PPI therapy, minimizing potential adverse effects on the gut microbiome and improving patients’ overall wellbeing.
Diet’s Role in Modulating Drug Effects
The influence of diet on the gut microbiome is significant, and it can affect how drugs work in the body. A diet high in fiber tends to promote a diverse and stable gut microbiome. Certain foods, especially those rich in probiotics, can foster the growth of beneficial bacteria while outcompeting harmful strains. This bactericidal competition may enhance the effectiveness of various medications and reduce side effects associated with dysbiosis. Studies support the idea that individuals who consume a Mediterranean diet experience better overall health and more effective drug absorption. For instance, higher intake of fruits, vegetables, and whole grains is linked to improved gut health, which can aid in metabolizing medications effectively. Understanding the synergy between diet and pharmacotherapy can lead to more tailored treatment plans. Healthcare providers ought to emphasize dietary considerations when discussing medication regimens with patients. Encouraging healthier eating habits could very well enhance drug effectiveness while reducing the chance of harmful side effects, thus improving patient outcomes. It stands to reason that integrating dietary advice into clinical practice might be a beneficial strategy for managing medications and gut health.
Research continues to illuminate the complexities of the interaction between medications and the gut microbiome. With the rise of precision medicine, understanding these relationships is increasingly important in tailoring treatment strategies to individual patients. The bidirectional nature of this interaction implies that healthcare providers must consider both the effects of medications on gut flora and vice versa. Ongoing research is critical to unravel the specific pathways and mechanisms involved in these interactions but also to develop guidelines for optimal medication management. Practicing integrative approaches by combining pharmacological treatment with lifestyle modifications may enhance therapeutic outcomes. In this context, patient education remains paramount. Patients should be informed of how their medications may affect their gut health and encourage them to adopt dietary changes that may promote a healthy microbiome. Awareness and proactive measures could therefore contribute significantly to overall health and wellness. As our understanding of the gut microbiome expands, healthcare professionals will be better equipped to support patients in navigating these complex interactions, ultimately leading to improved health outcomes and medication efficacy. Enhanced collaboration between researchers, clinicians, and patients will pave the way for future advancements in this essential field of health.